New York State Medicaid Update - March 2026 Reimbursement Change for New York State Medicaid Fee-for-Service Professional Drug Claims Special Edition Volume 42 - Number 3
In this issue …
Maximum Reimbursable Amount
Definitions and Resources
Claim Submission Requirements
PAD Clinical Criteria
PAD Search Tool
PAD Waste
PADs Obtained at No-Cost
Claims Edits
Questions and Additional Information
Practitioner-administered drugs (PADs) are covered outpatient drugs in accordance with §1927(k)(2) of the Social Security Act, and are typically administered in the office of the practitioner or other outpatient clinical setting. In accordance with New York State (NYS) Social Services Law 367-A, NYS Medicaid fee-for-service (FFS) will be changing the pricing benchmarks used to determine the maximum reimbursable amount for drugs provided and claimed separately by medical practitioners. As previously communicated in the Reminder: Upcoming Changes to Billing Requirements for New York State Medicaid Fee-for-Service Professional Drug Claims article published in the December 2024 issue of the Medicaid Update, NYS Department of Health streamlined the process of pending claims for manual pricing "By Report" ("BR"). Medicare and third-party liability secondary claims are excluded from these methodology changes. This guidance does not apply to claims submitted to a Medicaid Managed Care (MMC) Plan.
Maximum Reimbursable Amount
The NYS Medicaid program will reimburse for drugs furnished by practitioners to their patients using the benchmarks listed below (Table 1). The Maximum Reimbursable Amount (MRA) for drugs billed via Professional claim types will be determined by the National Drug Code (NDC) reported on the claim instead of the Healthcare Common Procedure Coding System (HCPCS) code*. Providers are required to report the NDC for all PADs billed by a private practitioner to NYS Medicaid and for all ordered ambulatory claims billed by clinics. For all drugs furnished in this fashion, it is expected that the practitioner will maintain auditable records of the actual itemized invoice cost of the drug, including the numbers of doses of the drug represented on the invoice. If the NDC is not referenced on a claim, NYS Medicaid will not provide reimbursement for the drug. Please note: Both the HCPCS units and NDC quantity must be reported separately and must reflect their respective dosage forms.
*Please note: In reference to PADs, HCPCS Level II 'J-codes,' which are alphanumeric codes beginning with the letter 'J' or, in the case of biosimilars, 'Q,' are the specific, standardized codes used to report injectable or non-oral medications. While HCPCS represents the broader coding system (HCPCS), in the context of professional drug claims, J-codes, Q-codes, and HCPCS codes are commonly used interchangeably to identify the drug product.
Table 1
| Drug Type | If National Average Drug Acquisition Cost (NADAC) is available, reimburse at: | If NADAC is unavailable, reimburse at: |
|---|---|---|
| Generics – Multi-Source | Lower of NADAC, Federal Upper Limit (FUL), State Maximum Allowable Cost (SMAC), or Acquisition Cost | Lower of Wholesale Acquisition Cost (WAC), FUL, SMAC, or Acquisition Cost |
| Brands – Sole or Multi-Source | Lower of NADAC, FUL, or Acquisition Cost | Lower of WAC, FUL, or Acquisition Cost |
| 340B Purchased Drug* | 340B Acquisition Cost | 340B Acquisition Cost |
*When claim is eligible for submission as a 340B purchased drug claim, providers should refer to the NYRx Medicaid Pharmacy Program – Pharmacy Manual Policy Guidelines (4.0 Basis of Payment, 340B Pharmacy Drug Claims).
Please note: Prices used by NYS Medicaid FFS are provided by First Data Bank.
Definitions and Resources
National Average Drug Acquisition Cost
Prices are a national pricing benchmark for prescription drugs, calculated by the Centers for Medicare & Medicaid Services (CMS) from a voluntary, monthly survey of retail pharmacies. NYS Medicaid does not have the ability to adjust pricing reimbursement structure for National Average Drug Acquisition Cost (NADAC). Providers should refer to the following contact information for NADAC pricing:
NADAC Help Desk
Toll-Free: (855) 457-5264
Email: info@mslcrps.com
Fax: (844) 860-0236
Federal Upper Limit
The Federal Upper Limit (FUL) is a price ceiling determined by the Secretary of Health and Human Services and used by CMS to control prices for certain medications.
State Maximum Allowable Cost
The State Maximum Allowable Cost (SMAC) is a maximum reimbursement limit that may be applied when determining the cost of multi-source generic drugs. Prime Therapeutics™ determines the SMAC for NYRx prices. For questions concerning a SMAC price, practitioners may complete a NYRx the Medicaid Pharmacy Program – State Maximum Allowable Cost (SMAC) Price Research Request Form. Please note: Only claims that use this benchmark are subject to appeal.
Wholesale Acquisition Cost
The Wholesale Acquisition Cost (WAC) is the estimated manufacturer list price for a drug to wholesalers or other direct purchasers, not including discounts or rebates. The price is defined by federal law.
Average Manufacturer Price
The average price that a manufacturer sells drugs to wholesalers or directly to retail pharmacies. Manufacturers must report Average Manufacturer Price Average Manufacturer Price (AMP) data to CMS.
340B Ceiling Price
The 340B ceiling price is the maximum price a drug manufacturer can charge a 340B entity for a covered outpatient drug. It is also the maximum price a Covered Entity should submit to the NYS Medicaid program for a 340B-acquired covered outpatient drug. State Medicaid programs must reimburse drugs at their actual acquisition cost and no greater than the established 340B ceiling price. The 340B ceiling price is statutorily defined as the Average Manufacturer Price (AMP) reduced by the rebate percentage, which is commonly referred to as the Unit Rebate Amount. Health Resources and Services Administration (HRSA) publishes ceiling prices to covered entities (CEs). Frequently Asked Questions (FAQs) on the 340B program, as well as information on how to ask additional questions, can be found on the HRSA "FAQs" web page.
340B-Acquired Drugs
Outpatient drugs purchased at significant discounts by eligible CEs under §340B of the Public Health Service Act must be identified to avoid duplicate discounts under Federal law 42 United States Code §256b(a)(5)(A)(i). All claims submitted directly to NYS Medicaid for 340B acquired drugs must be identified by including a UD modifier to the 340B drug line and be submitted at acquisition cost, inclusive of all discounts. NYS Medicaid will not accept a claim for a 340B drug when the cost of the drug is higher than the ceiling price. Please note: Medicare requires either the JG or TB modifiers be appended to the 340B drug line. NYS Medicaid will accept either JG or TB in place of the UD modifier for crossover claims.
Categories of Service
The above reimbursement methodology applies only to drugs claimed separately by practitioners. This update applies to the categories of service (COS) listed below:
- Free Standing or Hospital Based Ordered Ambulatory (COS "0163" for Clinic Based, COS "0282" for Hospital Based)
- Physician (COS "0460")
- Nurse Practitioner (COS "0469")
- Midwife (COS "0525")
- Podiatry Services (COS "0500")
Drugs administered in a facility or inpatient setting will continue to be reimbursed at the posted facility rates. This includes, but is not limited to, All Patient Refined-Diagnosis Related Groups (APR-DRG) and Ambulatory Patient Groups (APGs).
Claim Submission Requirements
National Drug Code
The National Drug Code (NDC) is a unique code that identifies a drug labeler/vendor, product and trade package size. The NDC and quantity of drug which was administered to the NYS Medicaid member must be included with all claims regardless of claim type. If no NDC is received, the NDC billed is invalid, the NDC billed is terminated, or the NDC/HCPCS combination is not associated to the HCPCS code, the claim may be rejected.
Quantity of Drug
In addition to the number of HCPCS units, the appropriate NDC and the quantity of drug administered to the NYS Medicaid member pertaining to the NDC must be submitted on the claim. Please note: This amount may be different from the number of billed HCPCS units. Providers may utilize the eMedNY "Medicaid Pharmacy List of Reimbursable Drugs" web page, to verify NDC unit types. The NDC-quantity reported must be in the units stated under the "Basis of MRA" value from eMedNY "Medicaid Pharmacy List of Reimbursable Drugs" web page files. Providers should refer to the Practitioner Administered Drug Search Tool Now Available on the eMedNY Website article published in the March 2024 issue of the Medicaid Update, for detailed instructions on proper reporting amounts.
Drugs with Assigned or Unique HCPCS Code
Claims for PADs should be submitted electronically (837-P transaction) or via paper (claim form) through the Medical Assistance Health Insurance Claim Form (eMedNY 150003 form). All NYS Medicaid member and provider information must be included, along with a valid NDC/HCPCS combination. This policy applies to all Professional and Medicare Crossover claims for PADs.
Miscellaneous or Unclassified HCPCS Codes
Miscellaneous or unclassified HCPCS codes are used when a specific drug does not have an assigned HCPCS. Once a permanent HCPCS has been assigned to the drug, the provider must submit claims with the permanent HCPCS code. Any claims submitted for a miscellaneous code after a drug has been assigned a permanent HCPCS code will be rejected.
Miscellaneous or unclassified HCPCS code claims will continue to be billed via paper claim on a Medical Assistance Health Insurance Claim Form (eMedNY 150003 form). Manufacturer invoices are required showing the acquisition cost of the drug administered, including all discounts, rebates, and incentives, per program policy. In addition, providers are required to report the NDC and quantity. Please note: This quantity may be different from the number of billed HCPCS units.
PAD Clinical Criteria
Certain PADs may have clinical criteria, coverage parameters, or recommendations by the NYS Medicaid Drug Utilization Review (DUR) Board that must be met. Drugs with clinical criteria requirements will continue to be billed via paper. To learn more about the additional clinical criteria and billing guidance, practitioners should visit the NYS Department of Health "Policies and Billing Guidance for Practitioner Administered Drugs" web page.
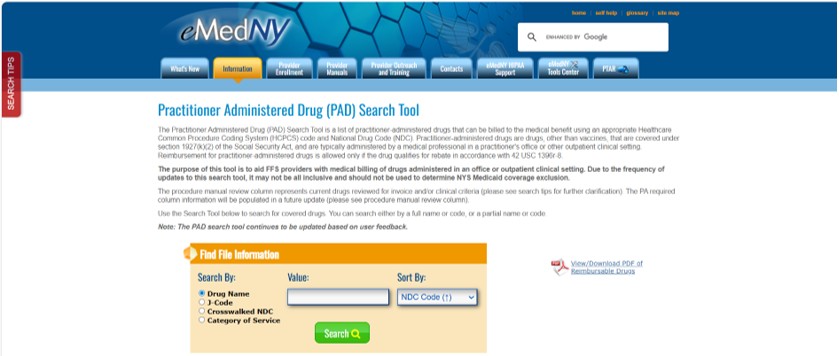
PAD Search Tool
The PAD search tool lists covered HCPCS codes for providers billing drugs separately and includes valid NDC/HCPCS combinations. The NDC/HCPCS must be listed accordingly on the crosswalk to be a covered benefit. The tool also aims to assist providers in utilizing the correct HCPCS and NDC billing units, as required by 2005 Federal Deficit Reduction Act. Information related to both the NDC unit type and HCPCS billing unit are displayed. Results can be expanded to view additional details of the HCPCS code.
Providers should utilize the red "SEARCH TIPS" tab on the left side of the eMedNY "Practitioner Administered Drug (PAD) Search Tool" web page for definitions and further descriptions. Providers should refer to the Practitioner Administered Drug Search Tool Now Available on the eMedNY Website article published in the March 2024 issue of the Medicaid Update, for detailed instructions regarding the PAD Search Tool and search results.
PAD Waste
NYS Medicaid FFS will reimburse providers for the unused, appropriately discarded, portion of a drug or biologic from a single-use vial or single-use package that is not administered to the patient. This reimbursement is only applicable to single-use vial/package. Drug waste from multi-use vials/packages will not be reimbursed. Payment will not be provided for an amount of drug that exceeds the maximum amount of drug or biologic as indicated on the single-use vial/package label.
Claims without Waste – JZ Modifier
The JZ modifier is required when there are no discarded amounts from single-use drug vials or single-use packages. All claims for single-use vials or single-use packages submitted where no waste occurs must have the JZ modifier (on the claim line with the administered amount) to attest that there were no discarded drug amounts. Providers administering a portion of a unit should round up to the nearest unit.
Claims with Waste – JW Modifier
The JW modifier is required when there is an amount of drug or biologic that is discarded from a single-use vial or single-use package after administering the drug to the patient. Providers must clearly report on a claim the number of HCPCS units of the drug administered and the number of HCPCS units wasted for a claim to be billable. All claims submitted for the discarded portion must include two separate lines. Line One is for the administered dose and Line Two is for the discarded amount. Information should be reported on two lines as follows:
- Line One: The HCPCS drug code and number of units administered. Providers administering a portion of a unit should round up to the nearest unit. If, through rounding up, the number of units reported on Line One equals the total number of units contained in the single-use vial/package, then Line Two is not required.
- Line Two: The HCPCS drug code appended with the JW modifier, and the number of units discarded or not administered to the patient.
Additional information on the use of the JW modifier may be found in the Clarification of Policy for Practitioner, Ordered Ambulatory, and APG Reimbursement -and- New Billing Instructions for Wasted Drugs Using Modifier JW article published in the August 2015 issue of the Medicaid Update. Paper claim submission and multiple drug procedure codes, including wastage, must be submitted for the same date of service on separate claim forms.
PADs Obtained at No-Cost
PADs that are dispensed by a pharmacy must be directly delivered to the site of administration. This is considered "white bagging" and is an acceptable practice. Drugs that are "white bagged" should be billed as a no-cost drug claim. Other examples of no cost drug could be drugs obtained via a patient assistance program, manufacturer samples, etc.
When a drug is "white bagged" the practitioner administers the drug that was delivered and obtained at no-cost as part of an office visit, the practitioner may only seek reimbursement for the administration of the no-cost drug. As per the Pharmacy Dispensing Drugs that Require Practitioner Administration article published in the December 2022 issue of the Medicaid Update, practitioners must bill the HCPCS code with NDC of the no-cost drug delivered to them by the pharmacy for administration, indicating a charged amount of $0.01, appended with the FB modifier (indicating the drug was supplied at no-cost). To be reimbursed for the administration of a no-cost drug, the practitioner must submit the applicable CPT code for the administration of the drug.
In some instances, a patient may bring a PAD to an office visit for education on self-administration or if the practitioner advises. This is considered "brown bagging." Brown bagging PADs causes concern regarding proper storage or handling, which can affect the efficacy of the drug. Brown bagging is acceptable only when the drug is intended, prescribed, or labeled for self-administration. Prescribers should use their professional judgement to determine the best method for NYS Medicaid members to obtain PADs. It is the responsibility of the pharmacist to ensure white or brown bagged drugs are appropriately dispensed. Practitioners will use the billing information above when administrating or training for the self-administration of the NYS Medicaid member or their caregiver, a drug that was dispensed directly to the NYS Medicaid member.
Providers should refer to the Clarification Regarding Pharmacy and Practitioner Dispensing of Drugs Requiring Administration by a Practitioner article published in the May 2023 issue of the Medicaid Update, for additional information on white and brown bagging.
Claims Edits
Providers needing assistance in understanding the reason for a NYS Medicaid FFS denied claim and possible solutions should refer to the eMedNY Edit/Error Knowledge Base (EEKB) Search Tool. The eMedNY EEKB Search Tool analyzes pending or denied claim responses returned on the following remittance statements: NYS Department of Health 835 X12 Remittance Advice Transaction or NYS Department of Health 277 X12 Claim Status Response. The eMedNY EEKB Search Tool returns detailed explanations about specific edits, including potential causes and possible solutions to resolve the problem. Providers should refer to the (Table 2) for a listing of new edit entries pertaining to this update. These new edits are intended to complement and enhance existing edits applied to HCPCS units and pricing benchmarks with the transition to NDC-based reimbursement structure.
| Edit # | Edit Description | Claim Adjustment Reason Code (CARC) | Healthcare Claim Status Code | Remark Code | Information and Resolution |
|---|---|---|---|---|---|
| 02365 | Claim Charge is Greater Than WAC Times Percentage | 16: Claim/Service lacks information or has submission/billing error(s) | 178: Submitted Charges | M79: Missing/Incomplete/Invalid Charge | The claim has a charge amount greater than the WAC reasonability amount. Resolution: Review the charge amount, units, procedure codes and modifiers submitted on the claim; correct and resubmit the claim if there are errors with any of these entries. |
| 02276 | Submitted Ingredient Cost Exceeds Ceiling Price | 16: Claim/Service lacks information or has submission/billing error(s) | 178: Submitted Charges | M79: Missing/Incomplete/Invalid Charge | The procedure code modifiers used on this PAD claim has flagged it as a 340B claim. The ingredient cost submitted for this 340B drug claim exceeds the ceiling price. Review the ingredient cost; correct and resubmit the claim if there is an error. Resolution: Review the ingredient cost, units and modifiers submitted on the claim. Resubmit if there is an error. Drugs not acquired through the 340B program should not be submitted with the 340B modifier. |
| 02275 | Invalid Codes Submitted for 340B Pharmacy Drugs | 16: Claim/Service lacks information or has submission/billing error(s) | 216: Drug Information | N823: Incomplete/Invalid Procedure Modifier(s) | The claim has a modifier billed that flags this drug as a 340B drug with a claim charge of zero. Resolution: Resubmit the claim with a charge greater than zero when using a 340B modifier code or resubmit the claim without the 340B modifier, if the drug was not acquired at a 340B price. |
Questions and Additional Information:
- NYS Medicaid FFS billing/claim questions should be directed to the eMedNY Call Center at (800) 343-9000.
- NYS Medicaid FFS drug policy questions should be directed to the Office of Health Insurance Programs Division of Program Development and Management by telephone at (518) 486-3209 or by email at NYRx@health.ny.gov.
- NYS Medicaid 340B policy questions can be sent to NYRx@health.ny.gov.
- Billing questions regarding the NYS Medicaid FFS program should be directed to the eMedNY Call Center at (800) 343-9000.
- Billing questions regarding MMC Plans should be directed to the plans.
The Medicaid Update is a monthly publication of the New York State Department of Health.
Kathy Hochul
Governor
State of New York
James McDonald, M.D., M.P.H.
Commissioner
New York State Department of Health
Amir Bassiri
Medicaid Director
Office of Health Insurance Programs